 Atoms of different elements differ in size, mass, and other properties. Atoms of a given element are identical in size, mass, and other properties. Atoms cannot be subdivided, created, or destroyed. 
The general tenets of this theory are: All matter is composed of extremely small particles called atoms. 
All atoms of the same element are alike and have the same mass. Dalton proposed his atomic theory in 1804. They cannot be divided into smaller particles, created, or destroyed. Atoms are the smallest particles of matter. Dalton’s atomic theory consists of three basic ideas: All substances are made of atoms. Rutherford needed to come up with an entirely new model of the atom in order to explain his results. From his research, Dalton developed a theory about atoms. In a famous quote, Rutherford exclaimed that it was "as if you had fired a 15-inch shell at a piece of tissue and it came back and hit you." No prior knowledge had prepared them for this discovery. John Dalton thought that all matter was made of tiny particles close particleA general term for a small piece of matter. Some were even redirected back toward the source. According to Dalton, the atoms of same element are similar in all respects. Parents: Joseph Dalton, Deborah Greenups. Born: Septemin Eaglesfield, Cumberland, England. Atoms are indivisible they cannot be broken down into simpler particles. Known For: Atomic theory and color blindness research. However an atom is the smallest particle that takes part in chemical reactions. Daltons atomic theory Everything is made up of very small particles called atoms. Surprisingly, while most of the alpha particles were indeed undeflected, a very small percentage (about 1 in 8000 particles) bounced off the gold foil at very large angles. The indivisibility of an atom was proved wrong: an atom can be further subdivided into protons, neutrons and electrons. The theory of atomism, proposed by Dalton in the early 19th century and derived from meteorological studies, is the foundation for our modern concept of the atom. 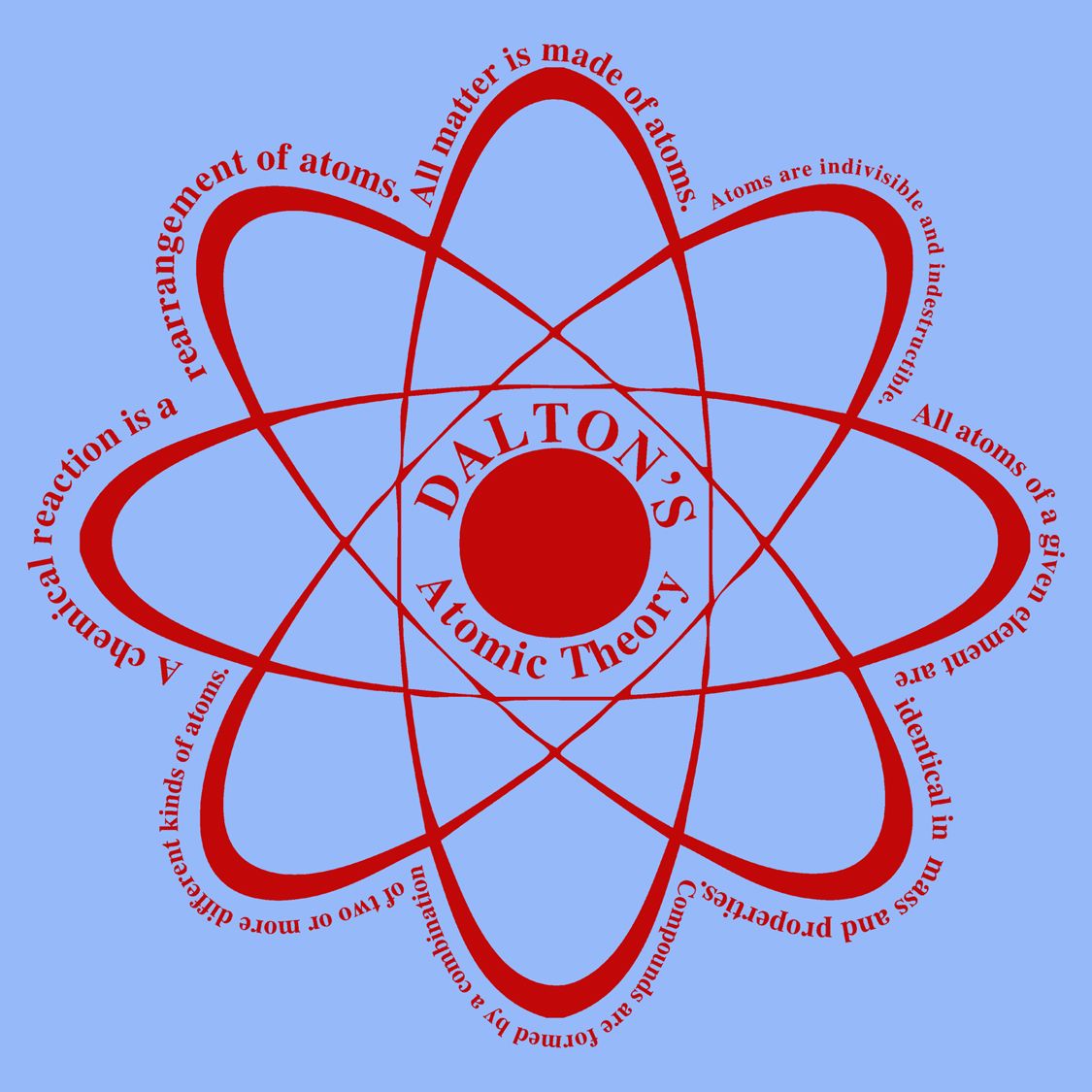
Rutherford found that a small percentage of alpha particles were deflected at large angles, which could be explained by an atom with a very small, dense, positively-charged nucleus at its center (bottom).Īccording to the accepted atomic model (Quantum Model), in which an atom's mass and charge are uniformly distributed throughout the atom, the scientists expected that all of the alpha particles would pass through the gold foil with only a slight deflection or none at all. (B) According to the plum pudding model (top) all of the alpha particles should have passed through the gold foil with little or no deflection. \): (A) The experimental setup for Rutherford's gold foil experiment: A radioactive element that emitted alpha particles was directed toward a thin sheet of gold foil that was surrounded by a screen which would allow detection of the deflected particles. Daltons atomic theory is the basis of the current atomic theory, though the atoms are no more considered ‘indivisible. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
